Tissue Engineering: Heart Valve Replacement
May 28, 2014
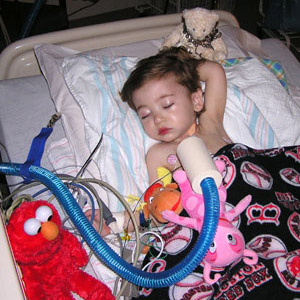
Dear Mr. and Mrs. Weglowski,
Thank you for your ongoing support with heart valve tissue engineering research at Boston Children's Hospital. Healing little Hearts, which has raised nearly $750,000 since 2008, enables us to continue our work to develop a living heart valve replacement. An important additional benefit is that these studies serve to elevate the standard of care for conditions affecting heart valves in all of our patients.
For those patients who need a complete valve replacement, there are many challenges to contend with. Artificial valves can cause blood clots and are not able to grow with the patient, requiring infants and children to face repeat operations as they mature. Bioprosthetic valves, which incorporate nonliving animal tissue, wear out with time. We think that the solution to these problems ultimately will come in the form of a tissue-engineered valve-a flexible and biodegradable scaffold onto which the patient's own cells are grown. A valve of this kind would have the potential to grow along with the child's heart, responding to signals from the rest of the body, living and adapting as a natural valve would.
With your support and hard work through Healing little Hearts, we are working towards freeing children with congenital defects from a lifetime of hospitalizations and surgeries. It is because of your philanthropy that this is possible.
Please accept my deepest thanks for helping us continue our work toward healing and sustaining the hearts of our patients through this research.
Sincerely,

John E. Mayer, Jr., MD
Top
February 2007
In a child's heart that beats 144,000 times a day, the valves, which ensure the blood flows in one direction only, are critically important for normal heart function. In many congenital heart defects, there are one or more valves that are either absent or malformed. These narrowed or absent valves impose an abnormal workload on the heart and in combination with other defects, may result in reduced oxygen levels. Congenital heart defects in which valves are absent or malformed include truncus arteriosus, tetralogy of Fallot, pulmonary valve stenosis, aortic valve stenosis, and certain forms of transposition of the great arteries. Although many of these defects can generally be addressed surgically with procedures which prolong the child's life, the surgical relief of narrowed valves often leaves residual valve leakage and replacement of a valve in a young child commits them to a series of valve replacement procedures to accommodate the child's growth. The currently available valve substitutes include an artificial heart valve, or a non-living or a chemically treated animal (bioprosthetic) valve. Neither solution is ideal: artificial replacement valves are more susceptible to infections and require blood thinner medications for life. The bioprosthetic heart valves have problems with limited durability, and these valves fail by becoming narrowed and/or developing leakage. Neither type of valve has any growth potential, committing young children who require valve replacements to multiple reoperations to place larger valves as the child grows.
It is for these reasons that cardiac surgeon John E. Mayer, Jr., M.D. has focused on developing a tissue engineering approach to create valve replacement tissue. The intent of these studies is to develop a process in animals in which cardiac valve tissue can be created from an animal's own cells (autologous) which are seeded onto biodegradable scaffolds. These scaffolds are used to provide initial spatial organization and strength for the developing tissue valve, but are designed to slowly dissolve so that a living valve structure remains. Experimental laboratory work has been in progress for 9 years, and experiments thus far have produced valves which have functioned in experimental animals for periods up to 8 months. Outstanding questions remain, however, including the choice of appropriate cell types for valve tissues, and development of optimal biodegradable scaffold materials. Recent work has focused on these two issues and has resulted in award-winning presentations at the national meetings of the American Heart Association and the Society of Thoracic Surgeons. These studies have shown that a population of cells that can be obtained relatively non-invasively from the circulating blood or from the bone marrow can be used to create tissues that seem to be usable for the creation of valve tissue. Former research fellows from Dr. Mayer's laboratory now lead research efforts of their own in the heart valve tissue-engineering field in Switzerland, Germany, and Japan.
Sincerely,
John E. Mayer, Jr., MD
Top
A step toward tissue-engineered heart structures for children
Delicate valves and patches grow with the child
September 10, 2007
 Infants and children receiving artificial heart-valve replacements face several repeat operations as they grow, since the replacements become too small and must be traded for bigger ones. Researchers at Boston Children's Hospital have now developed a solution: living, growing valves created in the lab from a patient's own cells.
In a special issue of Circulation published September 11, they describe making pulmonary valves through tissue engineering. These valves, which provide one-way blood flow from the heart's right ventricle into the pulmonary artery, are often malformed in congenital heart disease, putting an extra burden on the heart.
Infants and children receiving artificial heart-valve replacements face several repeat operations as they grow, since the replacements become too small and must be traded for bigger ones. Researchers at Boston Children's Hospital have now developed a solution: living, growing valves created in the lab from a patient's own cells.
In a special issue of Circulation published September 11, they describe making pulmonary valves through tissue engineering. These valves, which provide one-way blood flow from the heart's right ventricle into the pulmonary artery, are often malformed in congenital heart disease, putting an extra burden on the heart.
"The heart valve is a complex organ," says Virna Sales, MD, a researcher in Children's Department of Cardiac Surgery and the study's first author. "It must open and close synchronously, withstand pressure, and be pliable and elastic. We are one of the few labs in the U.S. that's attempting to make heart valves through tissue engineering. We hope these could just be implanted in a child just once, instead of the many heart operations most children have to go through as they get older."
The researchers, led by Sales and senior investigator John Mayer, MD, in Children's Department of Cardiac Surgery, first isolated endothelial progenitor cells (precursors of the cells that line blood vessel walls) from the blood of laboratory animals. They then "seeded" the cells onto tiny, valve-shaped biodegradable molds and pre-coated with proteins found in the natural "matrix" that surrounds and supports cells.
Experimenting with different matrix proteins and growth factors, they were able to make pulmonary valve leaflets that had the right mechanical properties -- sturdy yet pliable. Tests showed the original cells had differentiated to form both endothelial cells and smooth-muscle-like cells and added to the surrounding matrix to hold them together.
With grants from the American Heart Association and the Cambridge, Mass.-based Center for Integration of Medicine and Innovative Technology (CIMIT), Sales is now refining the lab-grown valves by exposing them to mechanical stress in a bioreactor. She is also using a "cardiac jelly" -- a cushiony material rich in matrix components and growth factors -- to encourage cells to differentiate and form a heart valve on their own, with only minimal reliance on an artificial scaffold. "I would like to mimic what really happens in the embryo -- what Mother Nature does," she says. The next step would be to implant the living valves into animals.
Sales and surgical research fellow Bret Mettler, MD, have already used tiny tissue-engineered patches in sheep to rebuild a portion of the pulmonary artery -- an area that often needs augmentation in patients with congenital heart disease. Eventually, Sales hopes to use tissue-engineering techniques to create "living stents" for adults with atherosclerosis. The current study was funded by the National Institutes of Health, a grant from the U.S. Department of Commerce's National Institute of Standards and Technology program (via Tepha, Inc., Cambridge, Mass.), the Gross Cardiovascular Fund, CIMIT, the National Science Foundation, and an American Heart Association National Scientist Development Grant awarded to Sales.
Contact:
Anna Gonski
617-355-6420
anna.gonski@childrens.harvard.edu
Boston Children's Hospital is home to the world's largest research enterprise based at a pediatric medical center, where its discoveries have benefited both children and adults since 1869. More than 500 scientists, including eight members of the National Academy of Sciences, 11 members of the Institute of Medicine and 10 members of the Howard Hughes Medical Institute comprise Children's research community. Founded as a 20-bed hospital for children, Boston Children's Hospital today is a 347-bed comprehensive center for pediatric and adolescent health care grounded in the values of excellence in patient care and sensitivity to the complex needs and diversity of children and families. Children's also is the primary pediatric teaching affiliate of Harvard Medical School. For more information about the hospital and its research visit: www.childrenshospital.org/newsroom.
For Donations by Check or Credit Card
• Donations by check can be sent to HEALING little HEARTS, P.O. Box 1535, Mattapoisett, MA 02739.
• You can also support the efforts of HEALING little HEARTS by making an online donation directly to Boston Children's Hospital by clicking the link below and following the prompts. Thank you!
Top


 All rights reserved.
All rights reserved.